


Currently Li-ion batteries dominate
the market share, powering electronic
gadgets and electric vehicles world
over, due to their high energy density
and design flexibility. However, the
scarcity of Li-reserves and rapidly
increasing demand for Li-ion batteries
for widespread applications strongly
defy sustainability. Accordingly, the
need for developing less expensive
and sustainable alternatives has
directed the focus towards the
upcoming Na-ion battery system;
especially in countries like India, which
is third largest producer of sodium.
However, there are certain
bottlenecks associated with the Na-ion
battery system. One among them is
the availability of a safe and
electrochemically stable anode
material; since graphite, the workhorse anode material in ‘conventional’
Li-ion batteries, does not work for Na-ion batteries.
A popular potential anode material is hard carbon, but which possesses issues
concerning
safety and irreversibility. The other bottleneck is the absence of a cathode
material which promises high
energy density, exhibits ‘long-term’ electrochemical stability and possesses
stability upon exposure to
air/water.
Against these backdrops, the NCPRE research team working under Prof. Amartya
Mukhopadhyay
have addressed both the above issues by leading to the development of a ‘bi-phase’
Na-titanate based
anode material and compositionally/structurally engineered high Na-containing
‘layered’
Na-TM-oxide (TM => transition metal) based cathode material.
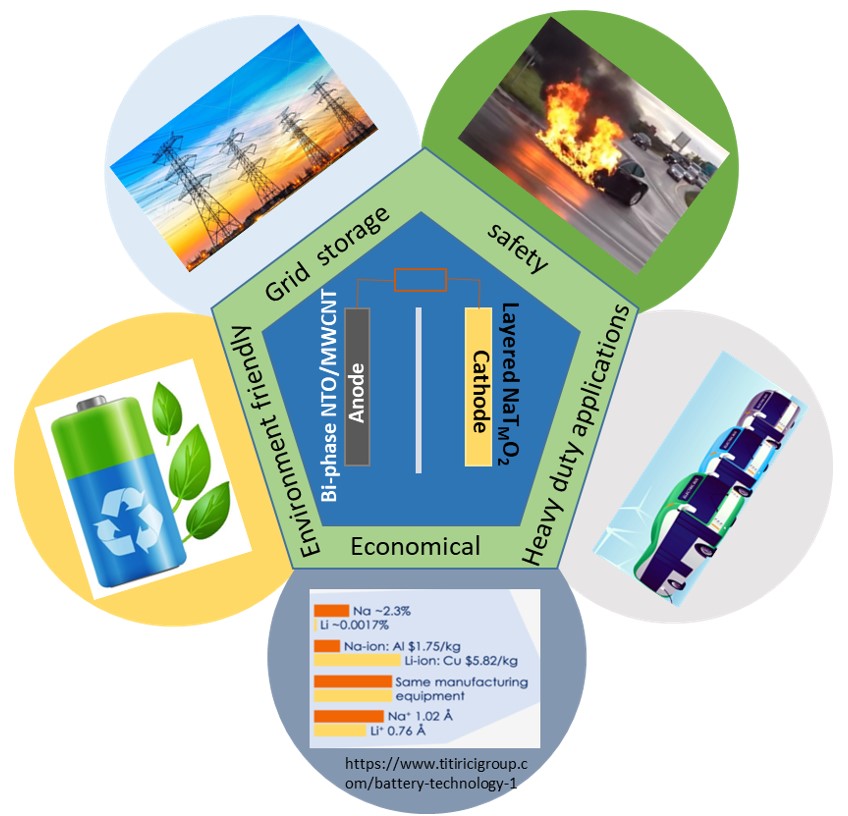
The ‘bi-phase’ Na-titanate based anode possesses a ‘safe’ operating potential, exhibits very long-term stability upon repeated discharge/charge and allows for extremely fast charging/discharging, so much so, >80% of its Na-storage capacity can be accessed in < 3 minutes. At the cathode front, our newly developed Na-TM-oxide not only exhibits very good long-term cyclic stability, but also inherently possesses excellent stability against degradation in the presence of moisture/water (unlike other variants); thus enabling electrode preparation via the health/environment-friendly and cost-effective aqueous route, which, in itself is a significant development for the Na-ion battery system.


